Plasma is the component of blood which contains the proteins that make blood clot.
The UK’s National Health Service Blood and Transplant (NHSBT) has embarked on a three-year project in collaboration with the Nonwovens Innovation and Research Institute (NIRI) and Macopharma, a supplier of blood packs and associated filters, to produce a ‘universal’ plasma filtration system.
Its success will mean patients who need transfusions won’t have to wait for blood-matched plasma.
Award
The project has been awarded £1.13 million in funding by the UK’s National Institute for Health Research (NIHR) which advances medical devices for increased patient benefit in areas of existing or emerging clinical need.
Plasma is the component of blood which contains the proteins that make blood clot and is often used to treat patients who suffer from severe bleeding after trauma, surgery or childbirth.
At the moment, a patient’s blood type must be compatible with the donated blood, or antibodies in the donor’s plasma (known as anti-A or anti-B) can attack the patient’s red blood cells causing anemia or even death.
In emergency situations, a patient’s blood type might not be known, so the safest option is to administer group AB plasma which is compatible with all blood types. However, only 4% of the population has AB blood so there are very limited supplies.
NHSBT is working with fellow researchers at NIRI and Macopharma to produce a filter that removes anti-A and anti-B antibodies from donated plasma so that anyone could receive a transfusion regardless of their blood group.
A universal plasma would speed up the process of getting it to patients who desperately need it and reduce the risk of patients having a reaction to the plasma, as well as simplifying its supply to hospitals and reducing the waste of valuable blood donations.
“Plasma transfusion is an essential treatment for patients with life-threatening bleeding,” said Dr. Gail Miflin, medical and research director at NHSBT. “Universal plasma would make it easier to ensure we can provide this in a timely and efficient way.”
A universal plasma would speed up the process of getting it to patients as well as simplifying its supply to hospitals and reducing the waste of valuable blood donations.
Hydrospace
Central to the development is a NIRI nonwoven technology called Hydrospace containing an active ingredient for the selective removal of harmful antibodies.
Hydrospace fabrics are lightweight hydroentangled fabrics with unique internal cavities within their cross sections. These can be of many different shapes and sizes and cavities smaller than 0.5 mm in diameter can be engineered into them. The cavities can be filled with gels, waxes, cosmetics, detergents and solid particles to functionalize the fabric.
Hydrospace nonwovens enable the storage and release or the controlled delivery of functional or active agents from the cavities. Fragile, hydrophilic and particulate materials can be introduced to the cavities during fabric formation. Adjacent spaces can be filled with different materials or the same spaces can be filled with dispersions, gels or combinations of solids and liquids as the fabric is made. The upper and lower surfaces of the fabric can be engineered in order to control the rate of delivery of the contents.
Hydrospace fabrics are lightweight hydroentangled fabrics with unique internal cavities within their cross sections.

Rubrovita
A second and related blood filtration project that Leeds, West Yorkshire-based NIRI is involved in is Rubrovita, which aims to develop a low-cost, blood separation unit which will enable donated whole blood to be split into its component parts.
This separation will allow a single donation of whole blood to be shared amongst several patients based on their required volume, but more importantly, allows the most appropriate component to be transfused to the patient.
In particular, the use of concentrated red blood cells (RBC) to treat anemia is far preferable to the use of whole blood, since most anemia patients have a normal blood volume and the transfusion of excess fluid volume may be detrimental to health.
In developed blood services, the separation of whole blood into component parts is primarily achieved by the use of centrifuges and semi-automated blood presses. It is envisaged that separation under gravity would benefit blood service providers in both developed and developing countries due to the simplified method of separation, but more importantly allow blood services in developing countries to provide the most appropriate blood components for patients – without the need for costly, complex equipment and an advanced infrastructure.
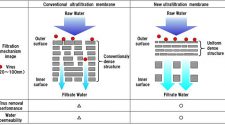
The Rubrovita project is now exploiting NIRI’s expertise in both nonwoven and membrane filtration technology. The use of membranes to separate red blood cells from plasma is not a new concept, but has largely been restricted either to small volumes for diagnostic assays or relatively costly units exploiting bundles of hollow fibers.
Bacteria and viruses
ExThera Medical Corporation, an early-stage medical technology company based in Martinez, California, and Vaals, The Netherlands, is meanwhile developing filters for reducing bacteria and viruses in blood that can lead to infections such as sepsis.
The company’s Seraph 100 Microbind Affinity blood filter is designed to reduce the duration and intensity of bloodstream infections to improve clinical outcomes. It is said to be the first and only broad-spectrum filter that reduces pathogens in whole blood.
As a patient’s blood flows through the Seraph Microbind Affinity Blood Filter, it passes over tiny microspheres coated with molecular receptor sites that mimic the receptors on human cells that pathogens use when they invade the body.
Harmful substances are quickly captured and adsorbed onto the surface and are thereby reduced in concentration in the bloodstream. Seraph does not add anything to the blood, which is returned to the patient with blood cells intact.
The adsorption media is a flexible platform using chemically-bound, immobilized heparin for its blood compatibility and its unique capacity to bind pathogens.
According to data revealed by the Centers for Disease Control and Prevention, sepsis affects around 800,000 people in the U.S. each year and it is the ninth leading cause of disease-related deaths in North America.
By using immobilized heparin with a unique molecular architecture, Seraph mimics the heparan sulfate binding sites on endothelial cells that are targeted by invading pathogens. Seraph’s high surface area quickly reduces large amounts of drug-resistant and drug-susceptible bacteria, viruses, toxins, and sepsis mediators in flowing blood.
Seraph may be configured with optional supplemental adsorbents to bind other toxins and evolved pathogens. For example, ‘Seraph 200’ adds endotoxin binding to Seraph 100, which may be useful in bloodstream infections that progress to endotoxemia. The Seraph filters have a blood-contacting surface that is anti-thrombogenic and anti-inflammatory.
“The need for advances in the treatment of bloodstream infections has, until now, gone largely unmet,” said Robert Ward, CEO of ExThera. “However, our technology has repeatedly shown promise in preclinical studies and clinical testing as a safe and effective option.”
ExThera is now working towards CE Mark approval for product introduction, initially into the EU, later this year.